The analysis of strontium isotopes is an increasingly common method in the toolkit of archaeology. Strontium analysis helps archaeologists understand where people lived in the past. It can provide insight about whether people were immigrants to an area or whether products like meat and wool were traded over long distances. Strontium analysis is being used to question such conventional wisdom as how Hyksos “invaders” took over New Kingdom Egypt and whether the famously nomadic Scythians were really so nomadic. But how does strontium analysis provide these insights? How does it work? What can it do, and – importantly – what can it not do?
All isotope analysis of biological materials works by exploiting the fundamental fact of the food chain. The food, water, and even air that animals and plants consume have chemical links to their environmental conditions. For example, in the radiocarbon analysis of plants, the carbon isotopes reflect the composition of the air when the plant was alive, and the slow breakdown of these isotopes after the plant has died allows for the plant to be dated. In oxygen analysis, factors like altitude and precipitation patterns affect local hydrological cycles, leading to different ratios of oxygen isotopes in drinking water.
Strontium analysis relies on the way the chemicals in soil and water derive from bedrock. Strontium has similar properties to calcium, so it can be substituted for calcium when living organisms build tissues like bone and tooth enamel. Strontium in bedrock is released when the bedrock weathers into soil, or it can get leached into the water that flows through or around the bedrock. The strontium then enters the food chain as plants draw nutrients from the soil and water where they grow, and it gets incorporated into human and animal tissues as they eat the plants and the animals that have been feeding on the plants.
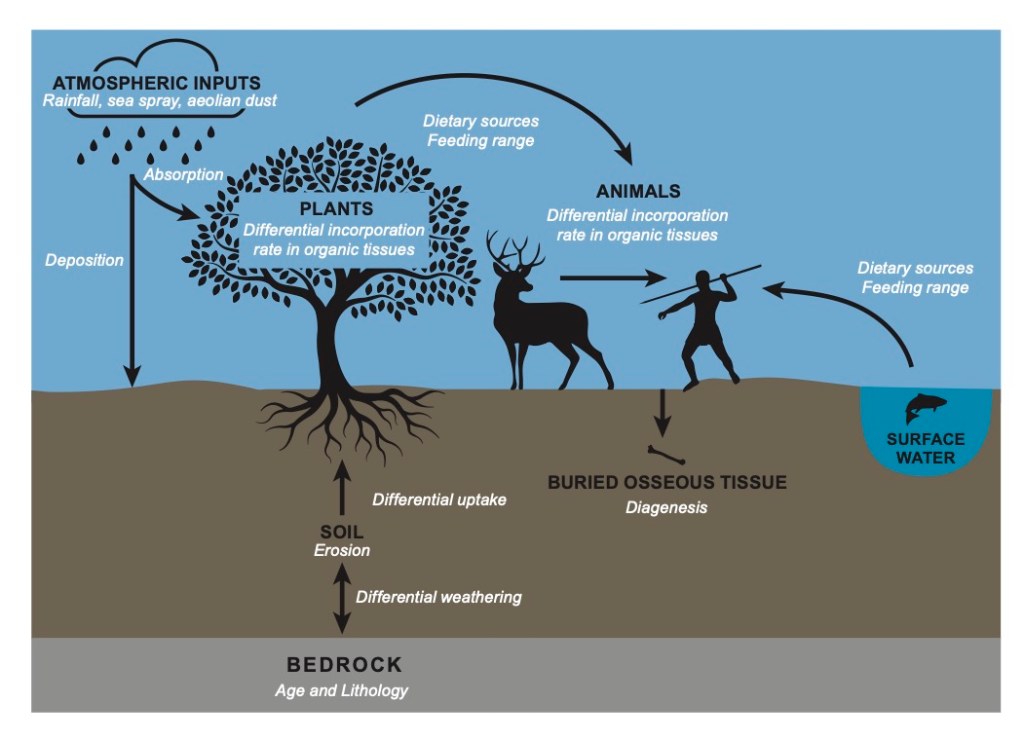
Strontium analysis wouldn’t work if all the strontium in all the bedrocks were the same, but helpfully it isn’t. Bedrocks have different ratios of strontium isotopes in them. Chemically, isotopes are atoms of an element that have the same number of protons but different numbers of neutrons. As an analogy, you can think of elements as ice cream and isotopes as their different flavors. Mint chocolate chip and butter pecan are both ice cream, but if I gave you a bowl with three scoops of mint chocolate chip and one of butter pecan, you’d have no trouble telling how much of each flavor was in the bowl. The element strontium has four flavors (isotopes) that occur commonly in bedrocks. Archaeologists can separate the strontium from a sample, and by looking at how much there is of one flavor (the 87Sr isotope) versus another flavor (the 86Sr isotope), archaeologists can describe the nature of the strontium in an area – its 87/86 strontium ratio.
Understanding a place’s strontium ratio lets archaeologists think about whether various organic tissues could come from that area. All kinds of tissues can potentially be used for strontium analysis, but the most common are bone and tooth enamel. Tooth enamel is particularly useful because it’s resistant to absorbing more strontium from the soil where it was buried – which could mess up the results of the strontium analysis – and because it forms when an individual is a juvenile. This means it can be very helpful for identifying when a person grew up in a different place from the one where they were buried, which is great for archaeologists who want to understand issues like migration, nomadism, and trade networks in the past.
Strontium analysis does have limitations. Similar strontium ratios can be found in many geographical regions, so strontium analysis is better at identifying where a person was not from than it is for pinpointing where they were from. Because strontium analysis works by excluding possible places of origin, it’s most useful when applied alongside other isotope analyses that can exclude additional places of origin. Another challenge of strontium analysis is that it can be difficult to understand whether the strontium ratio of soils in the present is an accurate reflection of the strontium ratios of the past. Modern fertilizers and other soil treatments can affect strontium ratios, so archaeologists have to be careful in using modern comparisons for ancient individuals.
Properly applied*, however, strontium analysis can be a powerful tool for addressing many of the enduring questions we have about the past, such as understanding the nature of ancient diasporas or reconstructing pre-modern globalism. I look forward to many more studies like the fascinating examples cited above.
* A summary of Holt, Evans, and Madgwick. 2021. Strontium (87Sr/86Sr) mapping: A critical review of methods and approaches. Earth-Science Reviews 216: 103593.